Surface tension is a fundamental physical phenomenon that governs how liquids behave at their interfaces. In simple terms, it is the elastic‑like pull that makes the surface of a liquid act as if it were covered by a stretched membrane. This effect arises from cohesive forces between molecules, which are strongest at the surface where molecules have no neighbors above them. By mastering the concepts behind surface tension, scientists and engineers can explain a wide range of everyday observations—from why water droplets form beads on a waxed car hood to how insects like water striders glide across ponds. In this article we will explore the molecular origins of surface tension, its practical implications, and how it is measured and controlled in modern technology.
What Is Surface Tension?
At the molecular level, each liquid molecule is attracted to its neighbors by intermolecular forces such as hydrogen bonding and Van der Waals interactions. Inside the bulk of the liquid these forces are balanced in all directions, but at the surface they are unidirectional, pulling inward. This creates a net force that minimizes the surface area, giving rise to the characteristic “tight” surface. The magnitude of this force is quantified as surface tension and expressed in Newtons per meter (N·m). The higher the surface tension, the more the liquid resists external force, which is why water—known for its relatively high surface tension—forms nearly perfect spheres when suspended in microgravity.
How Molecular Forces Create Surface Tension
The key driver behind surface tension is cohesion, the attractive force between like molecules. Cohesive forces are strongest when molecules are identical, as seen in pure water, but they can be altered by the presence of solutes or surfactants. Surfactants are compounds that reduce surface tension by disrupting the cohesive network at the interface; this principle is exploited in detergents, inks, and fire‑fighting foams. In addition to cohesion, the concept of surface energy—energy required to increase the surface area of a liquid—provides a thermodynamic perspective. When a liquid surface expands, work must be done against these molecular forces, which is why a droplet adopts a shape that minimizes surface area.
Everyday Examples of Surface Tension
Understanding surface tension helps explain many common phenomena. Below are several everyday examples:
- Rain droplets bead on a freshly waxed car because the wax lowers the surface energy of the glass.
- Insects such as water striders distribute their weight across a larger area, allowing them to stand on water without breaking the surface.
- Capillary action, the ability of a liquid to flow in narrow spaces without external forces, is driven by surface tension and adhesive forces; it enables plants to draw water from roots to leaves.
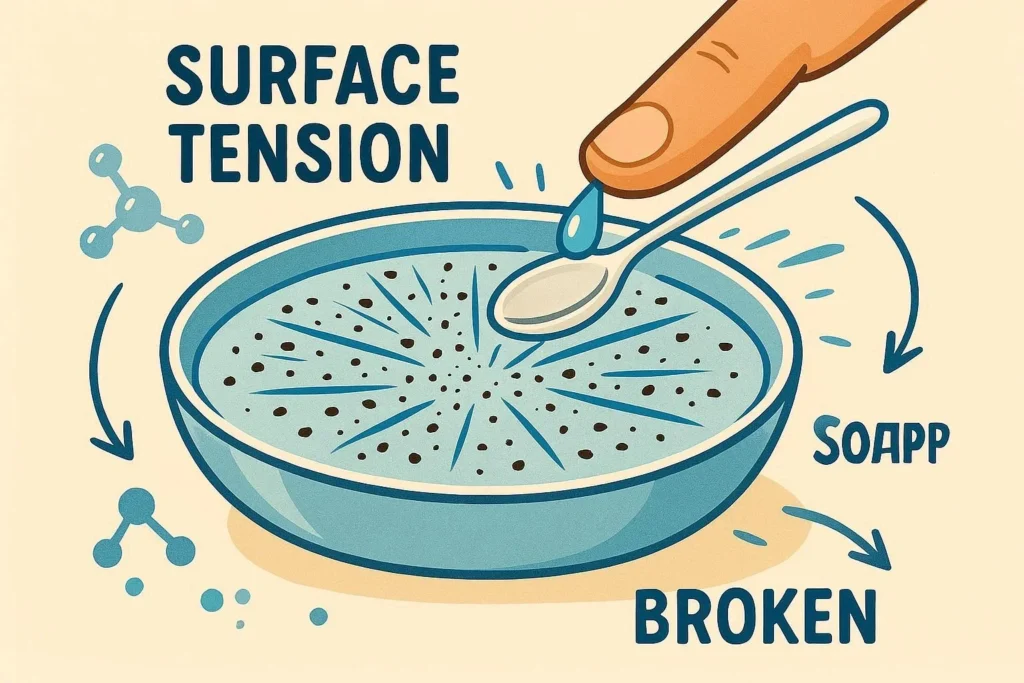
- When you sprinkle pepper on the surface of water and add a drop of dish soap, the sudden reduction in surface tension creates a vivid radial motion.
For a deeper dive into the physics of these phenomena, consult the Wikipedia entry on surface tension and the Britannica article on capillary action.
Measuring and Controlling Surface Tension
Scientists employ several techniques to quantify surface tension. The most common methods include the du Noüy ring method, the Wilhelmy plate technique, and pendant drop analysis. Each technique measures the force required to detach a probe from the liquid surface or analyzes the shape of a droplet under gravity. Accurate measurement is essential in industries such as pharmaceuticals, where surface tension affects tablet coating processes, and in aerospace, where fluid management in microgravity environments hinges on precise control of interfacial forces. NASA, for example, conducts experiments on the International Space Station to study fluid behavior in the absence of buoyancy, relying heavily on surface tension data (NASA fluid experiment).
When engineers need to modify surface tension, they often add surfactants or alter temperature, since surface tension decreases with rising temperature. The NIST reference database provides standardized values for a wide range of liquids across temperatures, serving as a valuable resource for research and quality control.
Why Surface Tension Matters in Modern Science
Beyond everyday curiosities, surface tension plays a pivotal role in advanced scientific fields. In microfluidics, the manipulation of tiny fluid volumes relies on surface tension to drive flow through channels smaller than a human hair. In medicine, the stability of emulsions—such as intravenous lipid solutions—depends on controlling interfacial tension with biocompatible surfactants. Moreover, environmental scientists monitor the surface tension of oil spills to predict spreading rates and inform remediation strategies.
By understanding and harnessing surface tension, professionals can design better products, improve manufacturing efficiency, and solve complex environmental challenges.
Ready to deepen your knowledge of fluid dynamics? Subscribe to our newsletter for the latest insights on surface phenomena, microfluidic innovations, and more—plus exclusive access to downloadable guides and expert webinars.
Frequently Asked Questions
Q1. What causes surface tension in liquids?
Surface tension arises from cohesive forces between molecules at the liquid’s surface. Molecules inside the bulk are pulled equally in all directions, but surface molecules experience a net inward pull because there are no neighboring molecules above them. This imbalance creates an elastic‑like film that minimizes surface area. The strength of this effect is quantified as surface tension, typically expressed in newtons per meter.
Q2. How does temperature affect surface tension?
Increasing temperature adds kinetic energy to molecules, which weakens the intermolecular attractions responsible for surface tension. As a result, surface tension decreases as temperature rises. This relationship is approximately linear for many liquids over moderate temperature ranges. Engineers often exploit this by heating fluids to lower surface tension during processes like coating or cleaning.
Q3. What are common methods for measuring surface tension?
The du Noüy ring method measures the force needed to detach a ring from the liquid surface. The Wilhelmy plate technique uses a thin plate that is pulled from the liquid, recording the capillary force. Pendant drop analysis examines the shape of a hanging droplet and calculates surface tension from its curvature. Each method offers advantages depending on the liquid’s properties and required precision.
Q4. Why do surfactants lower surface tension?
Surfactants are amphiphilic molecules that position themselves at the liquid‑air interface, disrupting the cohesive network of the liquid molecules. By inserting between surface molecules, they reduce the net inward pull, effectively lowering surface tension. This property is harnessed in detergents, inks, and foaming agents to improve spreading and wetting. The degree of reduction depends on surfactant concentration and molecular structure.
Q5. How is surface tension important in microfluidic technology?
In microfluidic devices, channels are often only a few micrometers wide, so surface forces dominate over gravity. Surface tension drives fluid motion through capillary action, enabling passive flow without pumps. Precise control of interfacial tension allows separation of droplets, mixing, and droplet generation in lab‑on‑a‑chip systems. Understanding surface tension is essential for designing reliable, low‑power biomedical and chemical analysis platforms.
Related Articles

100+ Science Experiments for Kids
Activities to Learn Physics, Chemistry and Biology at Home
Buy now on Amazon
Advanced AI for Kids
Learn Artificial Intelligence, Machine Learning, Robotics, and Future Technology in a Simple Way...Explore Science with Fun Activities.
Buy Now on Amazon
Easy Math for Kids
Fun and Simple Ways to Learn Numbers, Addition, Subtraction, Multiplication and Division for Ages 6-10 years.
Buy Now on Amazon